
1. Research Overview
Professor Hongjing Dou’s group at the School of Materials Science and Engineering, Shanghai Jiao Tong University, in collaboration with Professor Lefeng Qu and Associate Professor Kangkang Zhi from Changzheng Hospital’s Vascular Surgery Department, conducted research on supramolecular polysaccharide nanoparticles that function as platelet substitutes, mimicking the structure of platelets and surpassing their clotting function. These platelet substitutes can be assembled into macroscopic supramolecular materials that respond in wet environments by releasing the internal platelet substitutes and can be processed into various forms, suitable for different hemostatic scenarios. By synchronously assembling with blood cells and plasma proteins to form self-healing blood clots, the resulting hemostatic pad can rapidly form dynamic clots that adhere to tissues, achieving hemostasis in under 45 seconds in models involving liver and femoral artery bleeding in anticoagulated pigs. Furthermore, these materials can be removed without causing secondary bleeding or post-surgical adhesions, showing their potential to meet diverse hemostasis needs. The findings were published in Advanced Functional Materials under the title A Platelet-Substitute-Releasing Supramolecular Material for Cellular Assembly Mediated On-Demand Hemostasis (DOI: https://doi.org/10.1002/adfm.202422686) (Figure 1).
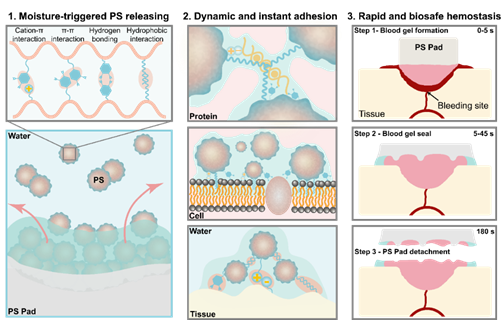
Figure 1. The platelet substitute pad rapidly releases its internal platelet substitute particles when triggered by water, assembling proteins, cells, and tissues to achieve efficient and safe hemostasis.
2. Research Background
With continuous advancements in hemostasis technology, various macroscopic hemostatic agents have been developed that can quickly stop bleeding by adhering to or filling wounds. However, these agents still face challenges addressing diverse post-surgical wound care needs. Although Materials at the micro- and nanoscale can form more precise and dynamic hemostatic clots that adapt to the microstructure of wounds, these materials can only programmatically interact with a limited range of cells or proteins, restricting the speed and dynamics of clot formation.
3. Main Research Findings
3.1 Adjustable Supramolecular Modifications Enable the Processing and Wet-Responsive Release of Platelet Substitutes
The group first used a polysaccharide containing amino and hydroxyl groups as a base and co-grafted catechol and alkyl groups onto the chain backbone to prepare a supramolecular polysaccharide conjugate. This conjugate self-assembles into nanoparticles as platelet substitutes in water, driven by hydrophobic interactions. The macroscopic pads, made from these nanoparticle aggregates, can be customized into various hemostatic forms for different bleeding scenarios, with the platelet substitutes rapidly released from the surface when triggered by moisture (Figure 2).
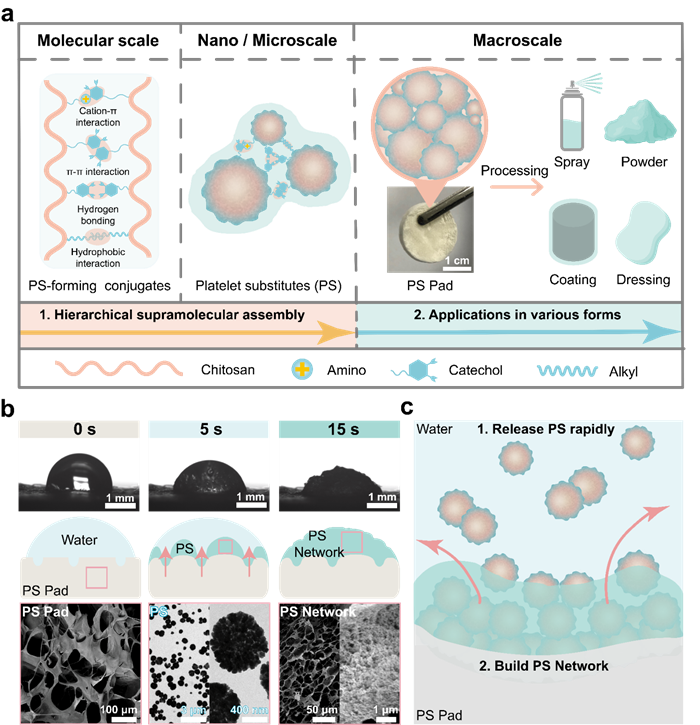
Figure 2. The processability and wet-responsive release characteristics of the platelet substitute pad.
3.2 Platelet Substitutes Facilitate Instant and Strong Blood Clot Formation
Different from the water contact angles that rapidly reduce, the platelet substitute pads maintain stable blood contact angles. The released platelet substitutes rapidly assemble with sufficient blood components, immediately forming a gel barrier that stops blood flow. The resulting blood gel also has self-healing properties, capable of withstanding up to 300% strain without being damaged, making it resistant to blood flow and external forces (Figure 3).

Figure 3. The coagulation performance in vitro of the platelet substitute pad.
3.3 Dynamic Multi-Scale Structure of the Blood Clots Assembled from Platelet Substitutes
In contrast to platelets, which stepwise and specifically bind to a small number of cells or proteins in the blood, the platelet substitutes released from the pads simultaneously assemble major blood components—red blood cells and albumin, forming a multi-scale composite structure. The red blood cells are tightly packed like bricks, forming micron-scale red blood cell walls, significantly enhancing the gel's modulus. The self-healing property of the blood gel results from the reversible supramolecular assembly between the platelet substitutes and red blood cells or albumin, providing the dynamic characteristics not seen in physiological blood clots (Figure 4).
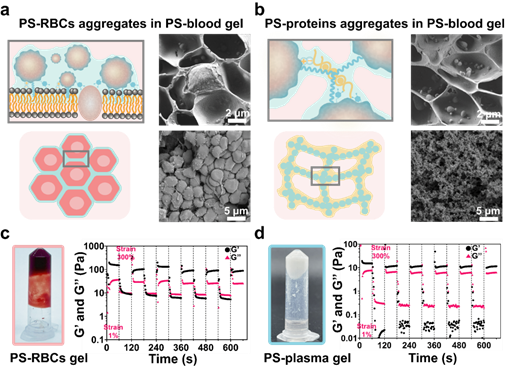
Figure 4. The multi-scale dynamic structure assembled by platelet substitutes with cells and proteins.
3.4 Instant and Stable Adhesion to Wet Tissues via Micron-scale Layers Formed by Platelet Substitutes
Unlike platelets, which can only adhere to specific cells or proteins, the wet-responsive release of the platelet substitute pads allows for immediate, non-specific adhesion to various vital organs, such as the heart, liver, spleen, lungs, and kidneys. The rapid formation of a micron-scale adhesive layer that adapts to the tissue's microstructure effectively prevents blood leakage from the wound interface and can withstand pressures exceeding human arterial blood pressure, demonstrating the potential for rapid hemostasis in high-pressure bleeding scenarios like arterial wounds (Figure 5).
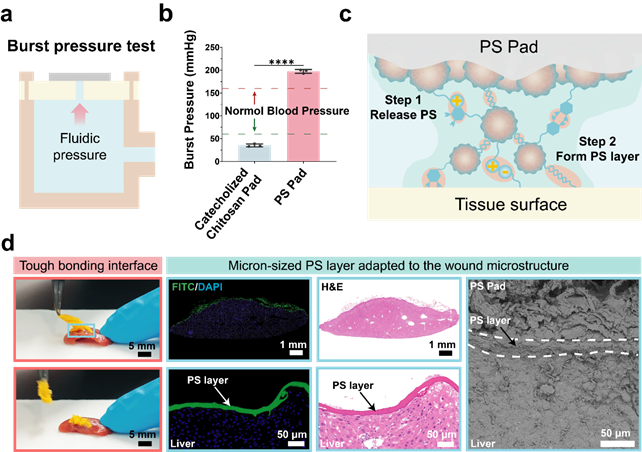
Figure 5. The wet tissue adhesion performance of the platelet substitute pad.
3.5 Rapid Hemostasis and Removal Without Rebleeding in Anticoagulated Pigs
In a simulation of clinical bleeding scenarios, the group injected heparin into 60 kg pigs to inhibit physiological clotting. In models with liver punctures and femoral artery punctures, the platelet substitute pads achieved hemostasis within 45 and 60 seconds, respectively. Compared to Johnson & Johnson's Fibrillar™ hemostatic pads, the platelet substitute pads reduced hemostasis time and blood loss by approximately 80% and 85%. Moreover, after stopping the bleeding, the pads could be rapidly removed without causing rebleeding, fulfilling the need for dynamic hemostasis in post-surgical wound care (Figure 6).
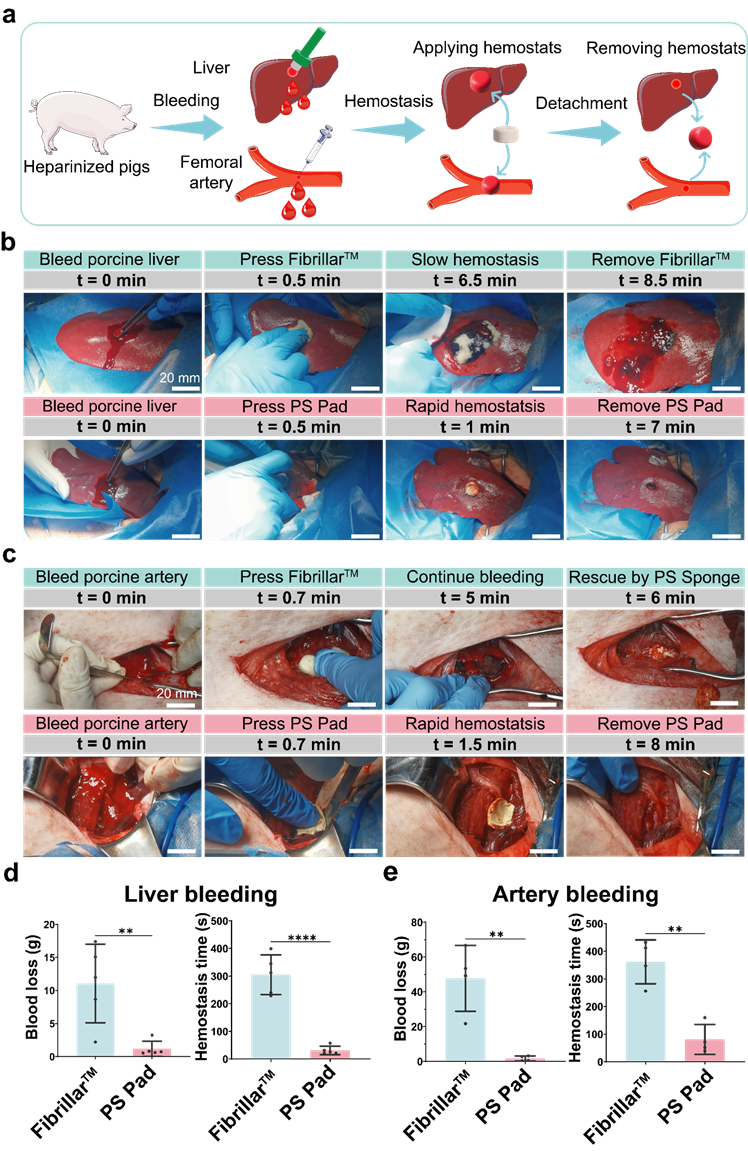
Figure 6. The hemostatic performance in anticoagulated pigs of the platelet substitute pad.
3.6 Prevention of Post-surgical Adhesion
Wounds that bleed tend to accumulate collagen, inevitably causing adhesion in the liver lobes. However, when using the platelet substitute pads for hemostasis, no adhesion or significant collagen deposition was observed in the liver lobes of rats, addressing the need for post-surgical adhesion prevention (Figure 7).
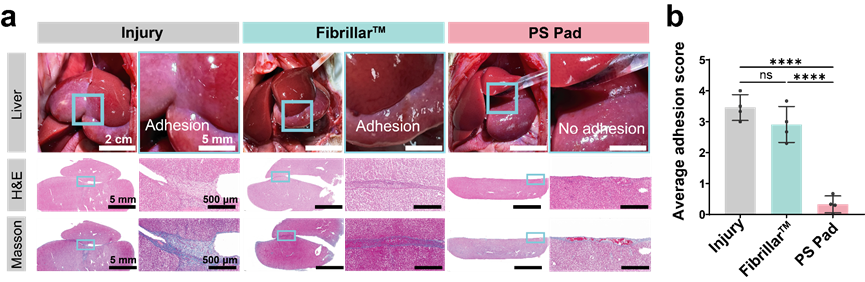
Figure 7. The anti-postoperative adhesion performance of the platelet substitute pad.
3.7 Mechanism of Rapid Hemostasis, Dynamic Removal, and Post-surgical Adhesion Prevention
The multifunctionality of the platelet substitute pad is derived from several key processes. Upon contact with blood, it rapidly releases platelet substitutes, immediately forming a blood gel barrier. In areas not covered by blood, the material quickly adheres to tissues by forming a micron-scale adhesive layer. The gel and adhesive layer on the tissue surface can dynamically detach from the non-blood-soaked patch and remain as a protective barrier at the wound site, preventing post-surgical rebleeding and tissue adhesion (Figure 8).
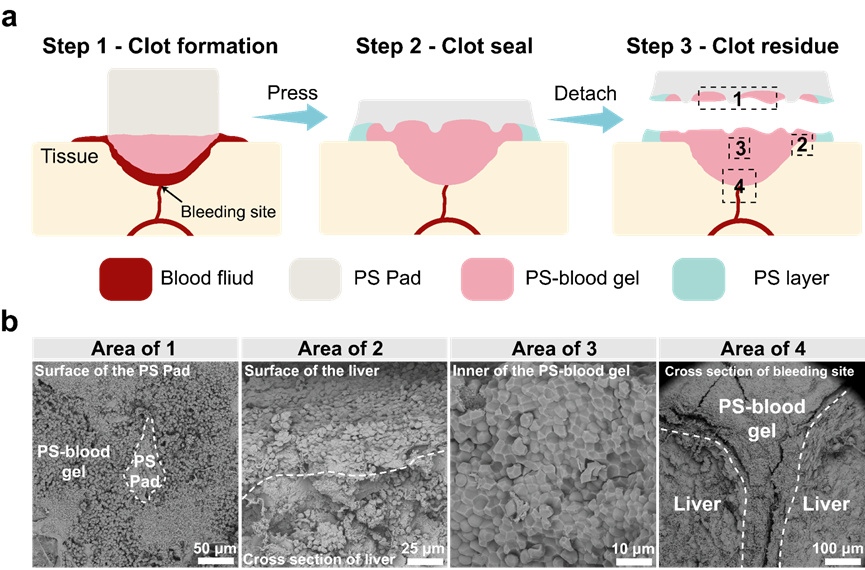
Figure 8. The hemostasis and anti-adhesion mechanisms of the platelet substitute pad.
4. Conclusion and Future Prospects
This research designs a multi-scale supramolecular material that releases platelet substitute nanoparticles, mimicking the microstructure of platelets while possessing non-specific binding capabilities to cells and proteins. This allows for the instant formation of multi-layered self-healing blood clots. It provides a new strategy for rapid and dynamic hemostasis, with promising applications in treating severe traumatic hemorrhages.
Ph.D. student Yirong Zeng from the School of Materials Science and Engineering, Shanghai Jiao Tong University, is the first author. Assistant Researcher Shuo Yang is the second author, and Professor Hongjing Dou from Shanghai Jiao Tong University, along with Professor Lefeng Qu and Associate Professor Kangkang Zhi from Changzheng Hospital, are the corresponding authors. This research was funded by the National Key R&D Program (2022YFE0100600), National Natural Science Foundation of China (21871180, 81971710, 82370498), National Translational Medicine Facility (Shanghai) Translational Medicine Research Fund (TMSK-2021-108, TMSK-2021-305), Shanghai Jiao Tong University "Jiao Da Star" Program (No. 20240304), and Changzheng Hospital's Innovation Clinical Research Project (No. 2020YLCYJ-Z09).
